[ad_1]
One piece of fine information for customers is that generic costs are falling. Nevertheless, generic costs could also be falling a lot that drug shortages are occurring (which isn’t a great factor). Knowledge from a working paper by Sardella (2023) finds a dramatic drop in generic costs lately.
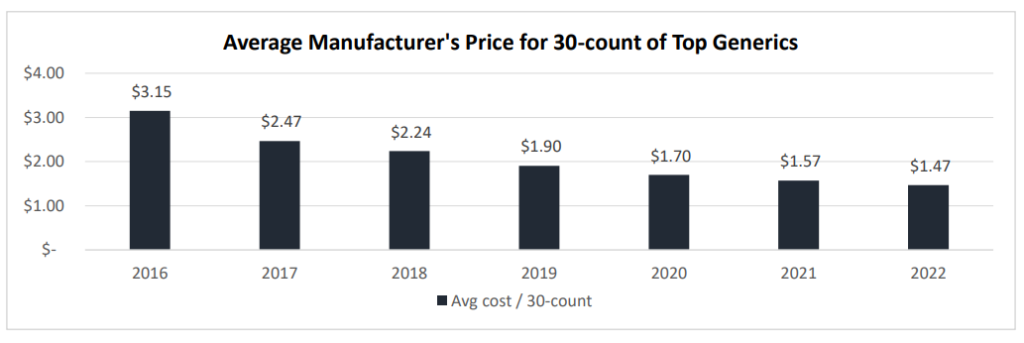
The authors declare that shortages in generic medicine are attributable to three major causes: (i) low profitability, (ii) low worth for high quality, and (iii) advanced, world provide chains.
With no distinguishing product differentiation or high quality monitoring [e.g., reputation] within the trade to differentiate product high quality variations, market competitors within the generic drug trade, with out market exclusivity, focuses on the dimension of worth.
Value competitors is particularly intense as a result of 3 massive pharmacy profit managers (PBMs) management 92% of the US market. Value competitors has lead most generic medicine are manufactured outdoors the US. Based on the FDA:
…as of August 2019, 72% of FDA-approved API manufacturing amenities have been outdoors of the US. A latest 2021 deeper dive revealed that roughly 75% of COVID-19 associated medicine, 97% of antibiotics, 92% of antivirals, and 83% of the highest 100 generic medicine consumed don’t have any US-based supply of APIs
International markets are engaging due to authorities subsidies, decrease prices of labor, and fewer regulatory oversight. Nevertheless, as a result of high quality just isn’t reimbursed, there are some points:
Higher than 80% of APIs for FDA-defined important medicines and over 90% of high antibiotics and antivirals don’t have any US manufacturing sourceLess than 5% of large-scale API websites, globally, are positioned within the US – the vast majority of large-scale manufacturing websites are in India and ChinaIndia and China have the best variety of API amenities supplying the US market and over ten p.c of those amenities have an FDA Warning Letter1
General, being a generic drug producer just isn’t an important enterprise. EBITDA (Earnings earlier than curiosity, taxes, depreciation and amortization) has fallen lately. Return on funding has fallen from near 10% in 2013 to only 5% in 2023.
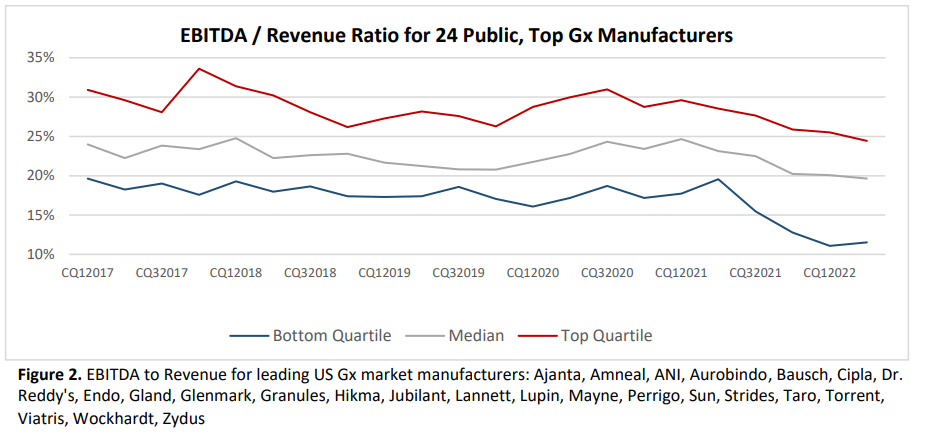
As a result of margins are so low, there’s little room to put money into high quality. Furthermore, compliance with FDA high quality requirements is falling.
…the speed of trade close-out of regulatory points (i.e., points resolved to the FDA’s requirements) has dropped from one-in-four warning letters closed out to one-in-twenty by 2022… 26% of the nation’s prescriptions now being equipped by corporations which have obtained warning letters since 2020.
The writer proposes 3 options to the issue which you’ll be able to learn right here.
[ad_2]
Source link



